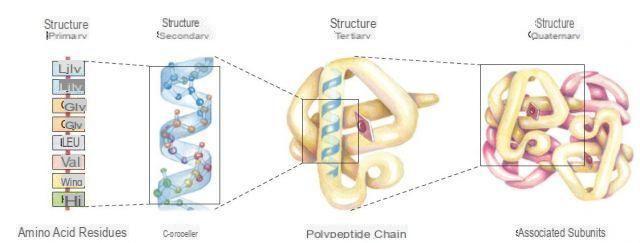
Introduction
Edited by Dr. Gianfranco De Angelis
The term "protein" is now widespread everywhere and everywhere we hear about protein supplements, protein powders, protein bars, protein drinks etc. However, few know its true meaning.
I will therefore try to explain what proteins are not from a biochemical point of view, but by highlighting their type of structure and relative classification.
Classification
Protein classification
- Based on the function performed: enzymes, hormones, transport, storage and structural proteins;
- According to the form: fibrous or globular proteins;
- On a chemical basis:
- simple proteins: composed only of amino acids
- compound or conjugated proteins: they are joined to other molecules (eg sugars, lipids, nucleic acids, metals, etc.).
Simple
There are two classes of simple proteins: fibrous and globular. The subdivision is based on the nature of the amino acids that are obtained with the hydrolysis of proteins, therefore their classification is based on the concepts of solubility and chemical composition.
Simple proteins give rise to eight groups of subdivisions: protamines, histones, albumins, globulins, glutelines, prolamins, phosphoprotides and scleroprotids.
Protamine
Le protamine they are rather simple proteins, since, by cleavage they give rise to only a few types of amino acids: mainly arginine and lysine. They therefore have a low molecular weight.
Soluble in water, of a strongly basic character, protamines are found in nature only in the animal kingdom, combined with nucleic acids to form nucleoprotides (especially in tissues with reproductive function, for example in the spermatozoa of many fish). They are not found free in nature
Protamines are free of sulfur amino acids, tryptophan and tyrosine; instead they are very rich in basic amino acids (in particular arginine).
Istoni
- istoni they are quite simple protides, even if more complex than protamines; like the latter, histones belong only to the animal kingdom, have a basic character and are soluble in water; they are distinguished instead for containing sulfur and for originating smaller quantities of exonic bases in the cleavage (arginine, histidine and lysine), of which they are however rich.
Like protamines, histones are not found free in nature, but combined with other substances to form protides. They are found in red blood cells, in leukocytes, in the heads of spermatozoa; globin is important, which constitutes the protein group of hemoglobin.
Albumin
Le albumin they are the best known and most easily accessible proteins; being very widespread, they gave their name to the entire class of protein substances (albumoid substances). In the animal kingdom they are widespread and mostly associated with globulins, as constituents of cells and cellular secretions. Some of their characteristic properties are: solubility in water, coagulability with heat and the possibility of giving rise to all amino acids by splitting (they are therefore complete proteins of good biological value). They have a high content of leucine (about 10-14%) and glutamic acid (7-13%); they also contain good quantities of arginine (6-10%) and lysine (6-8%).
The main animal albumin are: ovalbumin (or egg albumin) and whey albumin (or milk albumin). They are also found in many plants, although their properties are not perfectly known. A characteristic of animal albumin is to have a high content of sulfur, and a considerable percentage of amino acids cystine and methionine, while the vegetable ones contain modest quantities. Some plant albumins are poisonous; this is the case with ricin in castor oil.
Globuline
Le globulins they are the most common proteins, both in the animal and in the vegetable kingdom. They are very similar, from the point of view of the molecular constitution, to the albumins, but they differ in size. Furthermore, while albumin dissolve in water, globulins are non-polar; on the other hand they are soluble in neutral dilute saline solutions (NaCl). The most common are: blood globulins (α β, γ), lactoglobulin (milk), ovoglobulin (eggs), myosin and myoglobin (muscle). Vegetable globulins are found in particular in the seeds of many plants, especially in the oily ones of legumes; very rich in globulins are the proteins of soybeans and peanuts, where they form almost all of the protein substances. While animal globulins do not have major deficiencies in amino acids, those of the plant world are severely deficient in methionine (not surprisingly, the limiting amino acid of soy and other legumes).
Le Gluteline and prolamine (or gliadins) represent two groups of exclusively vegetable proteins, typically associated. Together, they make up the largest percentage of the cereal protein reserve (90-95%).
Gluteline
Le gluteline they have a fair amino acid balance and are very rich in glutamic acid, however present in lower concentrations than prolamines. They are insoluble in water, in saline solutions and in alcohol; they coagulate under heat and are soluble in diluted acids and bases. The gluteline of wheat, called glutenina, forms a protein complex with gliadin which constitutes gluten, essential for bread making and partly for the plasticization of flours. In rice, the gluteline present is called orizenine.
Prolammine o Gliadine
Le prolamine they contain, in considerable quantities, the amino acid proline, from which the name derives. They belong only to the vegetable kingdom, in fact, together with the glutelines, they constitute the most important part of many cereals, from which they are easily extracted. They are also called gliadins.
The best known gliadins are: the gliadin wheat, which gave its name to the whole class: the barley order; the zein of corn. They are insoluble in water and soluble in 60-80% alcohol. They do not coagulate in the heat.
Prolamines are rich in glutamic acid, which represents 20-30% of the amino acids of the seeds of cereals; proline and leucine are also abundant, while sulfur amino acids, lysine (which is not surprisingly the typical limiting aminoacid of cereals) and tryptophan (deficient in corn) are scarce. These amino acid deficiencies are responsible for the low protein efficiency of cereals. Congenital intolerance to gliadin is known as celiac disease.
Phosphoproteins
I phosphoproteins they are proteins particularly rich in phosphoric acid, therefore rich in phosphorus in the form of ortho-phosphoric acid, bound to esterify the alcoholic group of amino acids (such as serine). They have acidic properties due to the hydrogens of phosphoric acid which does not participate in esterification. Phosphoproteins should not be considered as conjugated proteins, nor should they be confused with nucleoproteins, capable of hydrolysis of phosphoric acid. Phosphoprotides are mainly present in proteins of animal origin, where there are two important representatives: the caseins of milk and the yolk of the egg yolk (the vitelline is one of the fundamental substances of the yolk and is a protein particularly rich in phosphorus). Ittulin of fish eggs is also remembered. The main constituents of these proteins are gluttamic acid (15-20%), serine (which abounds mainly in egg proteins), proline (5-10%) and lysine (5-7%). On the other hand, cystine is scarce.
Scleroprotidi
- scleroprotidi (o scleroproteine) are also called albuminoids or proteinoids and elusively belong to the animal kingdom. Unlike the proteins mentioned above, they are not part of the protoplasm. They are substances with considerable chemical resistance, insoluble in water and common solvents, which dissolve only in acids and even resist most proteolytic enzymes. By virtue of their exceptional chemical resistance, they perform mechanical functions of coating, protection and support, while they have little nutritional value. The most important scleroprotids in animal organisms are: collagen (fundamental constituent of connective, cartilage and bone tissue), elastin (basic constituent of the elastic fibers of tendons and vessel walls) and keratins (constituents of nails, hair and hair, but also scales, horns and feathers). Scleropeptides are composed of few amino acids: cystine (therefore sulfur) abounds in keratin, while collagen is rich in glycine (25%), proline and hydroxyproline, and deficient in sulfurates, tryptophan and tyrosine. Glycine and leucine are abundant in elastine, while cystine is scarce. Keratin is not attackable by gastric juices, therefore digestibility and intestinal absorption are very low; it derives a scarce importance in food. Boiling collagen with diluted acids increases its digestibility by transforming it into gelatin.
Conjugated Proteins
After a brief overview of the eight groups that subdivide simple proteins, it is necessary to speak, albeit briefly, of the second class of proteins, namely the conjugated proteins. They consist of a simple protide chemically joined to a non-protein group called prosthetics. Prosthetic groups are natural organic substances that act as enzymatic cofactors (helpers). The simple protein part is usually a protein from the histone or protamine group. The prosthetic group, on the other hand, is variable.
Depending on the type, four classes of conjugated proteins are distinguished:
- Glycoproteins (proteins with sugars)
- Lipoproteins (proteins with lipids)
- Phosphoproteins (contain phosphoric groups)
- Chromoproteins (contain metals, e.g. chlorophyll which contains Mg, or hemoglobin which contains iron)
The above description only wants to explain, albeit briefly, how proteins are classified, in order to make it clear that these substances, beyond their athletic function intended to "build" muscles, are fundamental for the life of all the organs of the our body, each of them having a specific task.